
Introduction
Many operations teams face a persistent challenge: magnesium chloride deicer fails unexpectedly in the field despite being rated to -28°F. The disconnect stems from a misunderstanding of eutectic temperature—the theoretical lower limit of ice-melting capability—versus the practical operational floor where MgCl₂ actually delivers useful performance.
While the eutectic point of magnesium chloride occurs at -28°F (-33°C) at 21.6% concentration, real-world effective performance typically drops off between 0°F and 5°F. Below this range, melting rates slow to impractical levels, and magnesium sulfate impurities crystallize in storage tanks and spray lines—failures that halt operations before the theoretical limit is ever reached.
This article breaks down the eutectic chemistry behind MgCl₂, explains what drives the gap between rated and real-world performance, covers how impurity crystallization triggers early system failures, and identifies when an alternative deicer is the better specification choice.
TL;DR
- MgCl₂ has a eutectic temperature of -28°F at 21.6% concentration—this is a lab value, not a practical working threshold
- Field effectiveness drops sharply below 5°F to 19°F depending on concentration, purity, and application method
- MgSO₄ impurities crystallize between 0°F and 5°F, clogging equipment before the eutectic limit is reached
- Product concentration, purity, and application method determine real-world performance far more than eutectic ratings alone
What Eutectic Temperature Means for Magnesium Chloride Deicer
The eutectic temperature is the specific temperature and concentration at which the solid-liquid phase boundary reaches its minimum—the lowest possible freezing point for that compound. For MgCl₂, this occurs at approximately 21.6% concentration and -28°F (-33°C).
Below this temperature, MgCl₂ brine begins to solidify rather than remain active, and ice-melting action ceases entirely regardless of application volume. The eutectic point functions as a governing constraint in deicer chemistry—a physical boundary of the system.
However, the eutectic temperature is a design-derived property, not an operational guarantee. The FHWA explicitly states that while MgCl₂ has a eutectic of -28°F, its practical effective working limit is much warmer. A product rated to its eutectic limit won't deliver reliable melting at that temperature in the field — it simply marks the point where the chemistry shuts down entirely. Real-world performance degrades well before that threshold.
Factors That Cause Real-World Performance to Deviate from the Eutectic
Three distinct failure mechanisms cause MgCl₂ to underperform before it ever approaches -28°F:
- Reaction rate collapse: Below approximately 5°F, the rate at which MgCl₂ brine melts ice drops to practically unusable levels. The product is still technically liquid, but melting action is negligible — the deicer fails in the field long before its theoretical limit.
- Impurity crystallization: Magnesium sulfate (MgSO₄) is a natural impurity in MgCl₂ sources, particularly Great Salt Lake brines. Unlike MgCl₂ itself, MgSO₄ crystallizes between 0°F and 5°F, forming sludge in storage tanks and clogging spray nozzles and transfer pumps. This failure is unrelated to the eutectic — it's a product purity problem.
- Field dilution: Precipitation, existing snow pack, and pavement moisture all reduce effective MgCl₂ concentration on the surface. As MgCl₂ melts ice or is diluted by snow and rain, the concentration decreases, causing the freezing point to rise back toward water — pushing the practical freeze point well above the rated eutectic value during the application window.

The Effective Operating Range of MgCl₂: Nominal, Boundary, and Safe Margin
Understanding where MgCl₂ performs reliably — and where it doesn't — comes down to three reference points: the nominal operating window, the boundary limits where performance drops off, and the safety margin operators should build into application planning.
Nominal Operating Range
Standard MgCl₂ liquid deicer (20–28% concentration) delivers reliable ice-melting performance above approximately 5°F to 19°F, with the strongest action between 15°F and 32°F. This temperature band covers most common winter maintenance operations.
Solutions below 20% active chemical have a meaningfully higher effective lower limit. Proprietary MgCl₂ blends with corrosion inhibitors may carry published effective lower limits as high as -4°F — a narrower working range than standard MgCl₂.
Allowable Tolerance and Boundary Limits
Upper boundary: MgCl₂ becomes relevant as a deicer at or below 32°F (0°C), but its hygroscopic activation advantage over solid salts begins to matter below about 25°F where dry sodium chloride becomes increasingly ineffective.
Lower practical boundary: Research and field experience consistently place the point of diminishing returns for MgCl₂ between 0°F and 5°F. The Minnesota Pollution Control Agency lists the lowest practical melting temperature for MgCl₂ as -10°F, while Missouri DOT field studies noted approximately -5°F as the effective floor. Below this range, melting rate is too slow for practical snow and ice removal, and equipment issues from impurity crystallization become a direct operational risk.
Safe Operating Margin
Plan a buffer above the lower boundary limit. Treating 5°F as a soft floor — rather than the theoretical eutectic of -28°F — keeps operations clear of the zone where melting performance and equipment reliability both deteriorate.
When temperatures are forecast to drop below 0°F:
- Switch to a higher-performance deicer chemistry (calcium chloride or a proprietary low-freeze blend)
- Increase pre-application rates so brine is already active before temperatures reach the failure zone
- Avoid relying on MgCl₂ alone as the primary deicing agent
Key Technical Properties That Govern MgCl₂ Effectiveness
Reaching -28°F in a lab setting is straightforward. Delivering that performance on an icy highway in January is not. Three physical properties determine how MgCl₂ behaves under real operating conditions: how it interacts with moisture, how concentration shifts its freeze point, and how impurities limit its working range.
Hygroscopicity and Brine Film Stability
MgCl₂ is hygroscopic — it pulls moisture directly from the atmosphere. At relative humidity as low as 35%, the compound draws enough moisture to initiate and maintain a thin brine film on pavement surfaces.
This makes it particularly effective as an anti-icer (pre-storm application). The brine layer resists initial ice bond formation without requiring moisture from melting, so it activates earlier than reactive deicers and holds the pavement surface longer.
The same hygroscopicity creates two operational risks worth knowing. In cold, dry conditions, continued moisture absorption can dilute the surface brine film over time, raising its effective freeze point.
On the warmer end, applying MgCl₂ to dry pavements above 28°F in high-humidity conditions can produce a dangerously slick surface film before any ice has formed — a risk that's often overlooked when anti-icing is treated as universally safe pre-treatment.
Concentration-Dependent Freeze Point
The freeze point of MgCl₂ solution is not fixed—it varies directly with concentration. At 21.6%, the freeze point is at its minimum (eutectic, -28°F). At lower concentrations, the freeze point rises substantially, meaning dilution from rain or snowmelt during application directly reduces the temperature range over which the product remains active.
Concentration-to-Freeze-Point Reference:
| Concentration (wt% MgCl₂) | Freezing Point |
|---|---|
| 10% | 17.9°F |
| 15% | 4.0°F |
| 20% | -17.3°F |
| 21.6% | -28.0°F (Eutectic) |
| 25% | -10.0°F |
| 28% | -1.0°F |
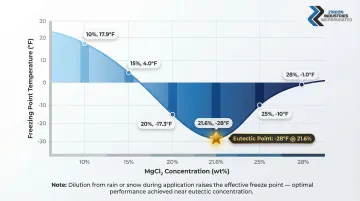
Liquid MgCl₂ products are typically supplied at 20–28% concentration because this range balances effective freeze-point depression with manageable viscosity and compatibility with spray equipment.
Impurity Sensitivity and Equipment Compatibility
MgCl₂ sourced from natural brines — particularly the Great Salt Lake — carries magnesium sulfate (MgSO₄) as a co-impurity. Unlike MgCl₂, MgSO₄ crystallizes at much higher temperatures, between 0°F and 5°F.
Once crystal formation begins, it blocks spray nozzles, clogs pump lines, and can immobilize liquid application systems entirely. The practical result: effective operational range drops well above the stated eutectic, regardless of the product's theoretical freeze point.
Higher-purity MgCl₂ formulations or products that include crystal-inhibiting additives extend the practical lower working limit by suppressing MgSO₄ crystallization. When sourcing liquid MgCl₂ for cold-weather operations, solution purity is a front-line procurement specification — not an afterthought.
How MgCl₂ Deicer Is Specified, Measured, and Applied in Practice
Commercial MgCl₂ deicer products are specified primarily by solution concentration (expressed as % by weight), which determines both the labeled eutectic point and the effective working temperature range. Standard liquid product is supplied at 20–28% MgCl₂; dry flake product contains 45–48% active chemical. Pacific Northwest Snowfighters specifications require a minimum of 25% MgCl₂ for corrosion-inhibited liquid magnesium chloride.
Operators should verify concentration on product datasheets rather than relying solely on eutectic temperature claims, since different concentrations carry meaningfully different working ranges.
Field measurement: Operators can verify concentration with a hydrometer or refractometer calibrated for MgCl₂ solutions, confirming actual concentration before filling spray equipment. Idaho Transportation Department specifies ASTM D1429 for specific gravity testing of brines. Cold storage can cause partial crystallization, altering effective concentration — so always verify before topping off spray equipment.
Automated liquid deicer spray systems eliminate measurement variability by delivering consistent, pre-set application rates regardless of operator judgment. In industrial environments — mining operations, conveyor systems, aggregate facilities — temperature windows are narrow and application rates matter:
- Under-treatment leads to equipment freeze-up and unplanned downtime
- Over-application wastes product and accelerates surface corrosion
- Automated systems remove the variability that manual spraying introduces
When temperatures drop below the practical limits of standard MgCl₂, automated delivery alone isn't enough — the chemistry itself has to change. Zircon Industries' Liquid Heat conveyor belt deicer, for example, is formulated to a -60°F freeze point to address conditions where standard MgCl₂ chemistry is insufficient.
Operating Beyond the Limits: Performance Loss, Equipment Issues, and Misinterpretations
Below 5°F, performance failure cascades across three failure modes:
- Melting rate drops to impractically slow levels for active snow and ice control
- Brine film may freeze in place rather than staying active on surfaces
- MgSO₄ impurities begin crystallizing in spray lines, causing downtime that halts operations
This is particularly critical in quarrying, mining, and road maintenance contexts where continuous equipment availability is non-negotiable.
The most common misinterpretation: citing the eutectic temperature (-28°F) as the product's working limit on datasheets or in procurement decisions. In reality, the eutectic is the phase-transition floor for a pure, controlled solution—not a field performance guarantee.
Operators who select MgCl₂ for environments regularly hitting sub-zero temperatures based solely on the eutectic claim will find the product underperforms well before reaching that threshold. For sustained performance below 0°F, purpose-formulated products with lower practical freeze points are the more reliable choice — particularly for mining equipment, conveyor belt deicing, and extreme-climate road maintenance.

That eutectic confusion isn't the only source of field failures. Two other misinterpretations regularly cause problems:
- Assuming that applying more MgCl₂ product compensates for temperature-driven performance loss—higher application rates cannot overcome the thermodynamic limit where brine solidifies
- Ignoring the interaction between temperature and concentration, leading operators to apply properly-rated product at the wrong dilution and then attribute performance failures to the product rather than the application method
At 20°F, calcium chloride flake melts 29% more ice after 10 minutes than MgCl₂ flake, and at 0°F, CaCl₂ melts 40% more ice after 5 minutes. When pavement temperatures drop below 15°F, agencies recommend switching to CaCl₂ or adding it to the brine mixture.
Frequently Asked Questions
At what temperature does magnesium chloride stop working?
MgCl₂ has a theoretical eutectic temperature of -28°F, but its practical field performance floor is 0°F to 5°F. Below this range, melting rate becomes too slow to be useful, and impurity crystallization can disable equipment long before the theoretical limit is reached.
At what temperature does magnesium chloride melt ice?
MgCl₂ delivers effective ice melting above 5°F to 19°F, with strongest performance between 15°F and 32°F. Concentration matters: dilution from snowmelt raises the practical freeze point significantly.
How effective is magnesium chloride at melting ice?
MgCl₂ performs well as both a de-icer and anti-icer above 5°F, and its hygroscopic nature lets it activate without external moisture — useful for pre-storm applications. Below 20°F, melting rate slows considerably compared to calcium chloride.
Which is better for melting ice, calcium chloride or magnesium chloride?
Calcium chloride has a lower eutectic (-60°F vs. -28°F), faster melting rates, and deeper ice penetration below 20°F. It also requires a lower application rate, making CaCl₂ the stronger performer in severe cold. MgCl₂ is a cost-effective choice in the moderate winter range above 5°F.
Is magnesium chloride safe for deicing?
MgCl₂ is lower in acute environmental toxicity than sodium chloride and produces less skin and surface irritation on contact than calcium chloride pellets. That said, it can cause severe concrete deterioration through reaction between Mg²⁺ and cement paste, and contributes chloride load to soil and waterways if over-applied.
At what temperature does deicer stop working?
The stopping point varies by deicer type: sodium chloride loses effectiveness around 15°F–20°F; magnesium chloride between 0°F and 5°F practically; calcium chloride maintains useful performance to around -25°F. For all deicer types, lab eutectic values are theoretical minimums — actual field performance floors are meaningfully warmer than those published limits.


