
Introduction
Over 20% of long-tenured miners in Central Appalachia now show radiographic evidence of black lung disease — and coal workers' pneumoconiosis (CWP) rates are still climbing despite decades of regulation. Effective dust suppression is no longer just an OSHA checkbox. It's a core operational and safety requirement.
Plain water sprays alone can't solve this problem. Coal's naturally hydrophobic surface repels water droplets, allowing fine respirable particles (those smaller than 4 microns) to slip through spray systems and reach miners' lungs. That's where surfactants come in. These surface-active agents transform water's physical properties, enabling it to capture the dangerous sub-micron particles that cause lung disease.
This guide covers how surfactants work at the molecular level, the three main surfactant types and their performance characteristics, key selection factors including coal type and water chemistry, and proven application methods that deliver measurable dust reduction.
TLDR:
- Surfactants improve overall dust capture up to 93% and fine-particle capture up to 125%
- Nonionic surfactants handle variable mine water conditions better than ionic types
- Dose near the critical micelle concentration (CMC); exceeding it wastes product with no added benefit
- Spray at 80-100 psi to generate fine droplets without excess air entrainment
- Foam systems cut dust 20-60% more than water sprays while using less water
Why Coal Dust Is a Serious Occupational and Environmental Hazard
The 1.5 mg/m³ Standard and Rising CWP Prevalence
MSHA enforces a permissible exposure limit (PEL) of 1.5 mg/m³ for respirable coal mine dust over a full work shift. Yet despite this regulation—which took effect in August 2016—CWP prevalence has climbed steadily since the late 1990s. Among underground coal miners with 25+ years of tenure, national CWP prevalence now exceeds 10%, and in Central Appalachia (Kentucky, Virginia, West Virginia), the rate reaches 20.6%.
The Sub-3 Micron Threat to Respiratory Health
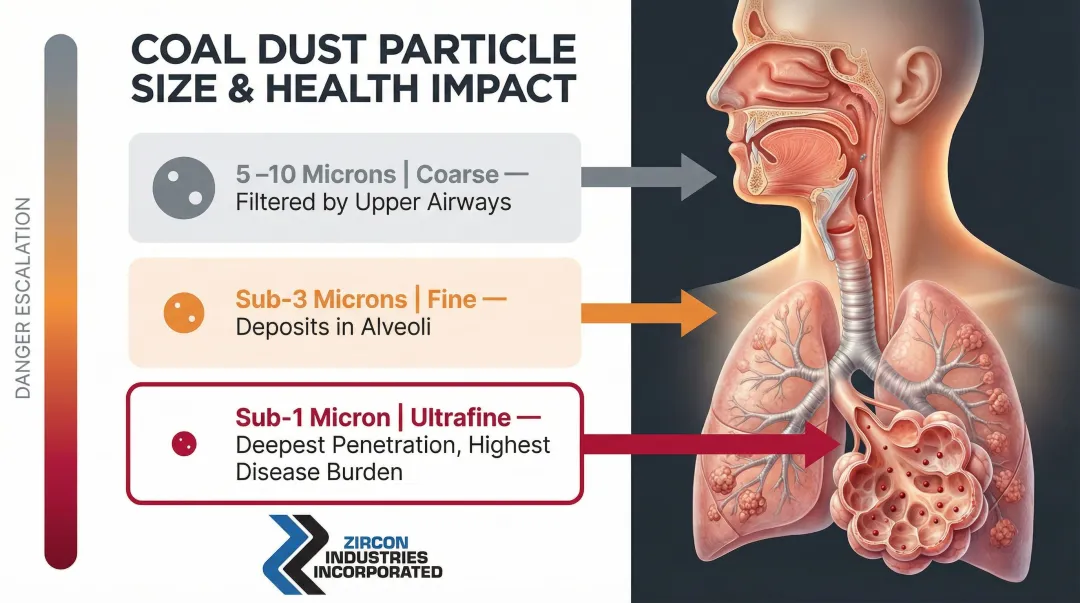
Respirable dust is defined by a 50% cut-point at 4 microns aerodynamic diameter, capturing particles generally 10 microns and smaller. But the real danger lies in particles below 3 microns. These ultra-fine particles penetrate deep into the alveolar regions of the lungs where gas exchange occurs, and they accumulate over time. Once embedded, they trigger inflammatory responses that lead to progressive lung tissue scarring—the hallmark of black lung disease.
Particle size determines disease severity far more than total dust mass. At equal concentrations:
- Coarse dust (5–10 microns): Largely filtered by upper airways before reaching the lungs
- Sub-3 micron dust: Bypasses airway defenses and deposits directly in the alveoli
- Sub-1 micron dust: Penetrates deepest, with the highest cumulative disease burden
Any suppression system that captures visible dust but leaves sub-3 micron particles airborne is failing where it matters most.
Explosion Hazard and Secondary Environmental Risks
The same fine particle behavior that makes coal dust a health threat also drives its explosive potential in underground mines. Float coal dust (particles passing a No. 200 sieve, <75 microns) reaches a minimum explosible concentration (MEC) of approximately 60 g/m³ for Pittsburgh seam bituminous coal. When suspended in air within this concentration range and exposed to an ignition source, coal dust can fuel catastrophic explosions.
Surface operations carry additional risks beyond explosion:
- Airborne dispersal contaminates surrounding communities and water bodies
- Abrasive particles accelerate wear on conveyor belts, seals, and mechanical equipment
- Settled dust on vegetation disrupts photosynthesis and local ecosystems
Why Plain Water Alone Falls Short for Coal Dust Control
Coal's Hydrophobic Surface Chemistry
Coal is inherently hydrophobic—its carbon-rich, non-polar surface actively repels water. When plain water droplets contact coal particles, high surface tension (approximately 72 dynes/cm at 20°C) prevents spreading. Instead, water beads up and rolls off, leaving particles dry and airborne.
This hydrophobic barrier occurs at the molecular level. Coal surfaces lack the polar functional groups (hydroxyl, carboxyl) that would form hydrogen bonds with water molecules, producing poor wetting, minimal agglomeration, and ineffective dust capture.
The Slip-Stream Effect: How Fine Particles Evade Capture
Even when water sprays directly intersect dust clouds, fine respirable particles (<4 microns) often escape capture through aerodynamic deflection. Larger particles (>10 microns) have sufficient inertia to impact spray droplets directly. But sub-4 micron particles follow airstreams around the droplet surface—a phenomenon called the "slip-stream" effect.
These sub-4 micron particles are exactly the ones that penetrate deepest into lung tissue—and they're the ones plain water is least equipped to capture.
The Two Core Problems Surfactants Solve
Effective coal dust suppression requires overcoming two physical barriers:
- Reducing water surface tension so spray droplets can spread across hydrophobic coal surfaces rather than beading off
- Converting coal particle surfaces from hydrophobic to hydrophilic so they become receptive to water agglomeration
Surfactant chemistry addresses both barriers at once—lowering surface tension from ~72 dynes/cm to as low as 30-35 dynes/cm while flipping coal's surface chemistry from water-repellent to water-receptive.
How Surfactants Work to Suppress Coal Dust
Molecular Structure: The Amphiphilic Design
Surfactants are surface-active agents built on an amphiphilic molecular structure—meaning they have dual chemical nature. Each molecule contains:
- Hydrophilic (water-loving) head group that bonds readily with water molecules
- Hydrophobic (oil/carbon-loving) tail that anchors to non-polar surfaces like coal
This dual nature allows surfactant molecules to orient themselves simultaneously at two critical interfaces: the air-water boundary and the coal particle-water boundary.
Primary Mechanism: Surface Tension Reduction
When added to spray water, surfactant molecules migrate to the air-water interface, positioning their hydrophobic tails toward the air and hydrophilic heads toward the water. This arrangement breaks up the hydrogen bonding that gives water its high surface tension.
Surface tension drops from ~72 dynes/cm to 25-30 dynes/cm. That reduction means water droplets spread across hydrophobic coal surfaces rather than bead off. The lower energy barrier allows water to wet particles that plain water cannot touch.
Secondary Mechanism: Surface Adsorption and Hydrophilic Conversion
Surfactants also attack the problem from the particle side. Molecules adsorb onto coal particle surfaces with their hydrophobic tails anchored to the carbon and their hydrophilic heads facing outward into the water phase.
This reorientation converts the coal surface from hydrophobic to hydrophilic. Once coated with surfactant, coal particles become receptive to water agglomeration and join into larger, heavier clusters that settle out of the air.
The Electrostatic Dimension: Charge-Based Capture
Both spray droplets and pulverized coal particles carry electrostatic charges. The type of surfactant influences droplet charge:
- Anionic surfactants create net negative droplets
- Cationic surfactants create net positive droplets
- Nonionic surfactants produce weakly positive or neutral droplets
When spray droplet charge opposes coal particle charge, Coulombic attraction enhances capture efficiency. When charges match, electrostatic repulsion reduces effectiveness. This charge dynamic explains why surfactant selection matters—especially when coal dust carries a strong consistent charge.
Proven Performance Gains from Research
Academic research by Jin Kim (1995) provides foundational data on surfactant efficacy. The most effective formulations delivered substantial gains over plain water across two key measures:
- Overall dust suppression: up to 93% more effective than plain water
- Fine particle capture (<3 microns): up to 125% more effective than plain water
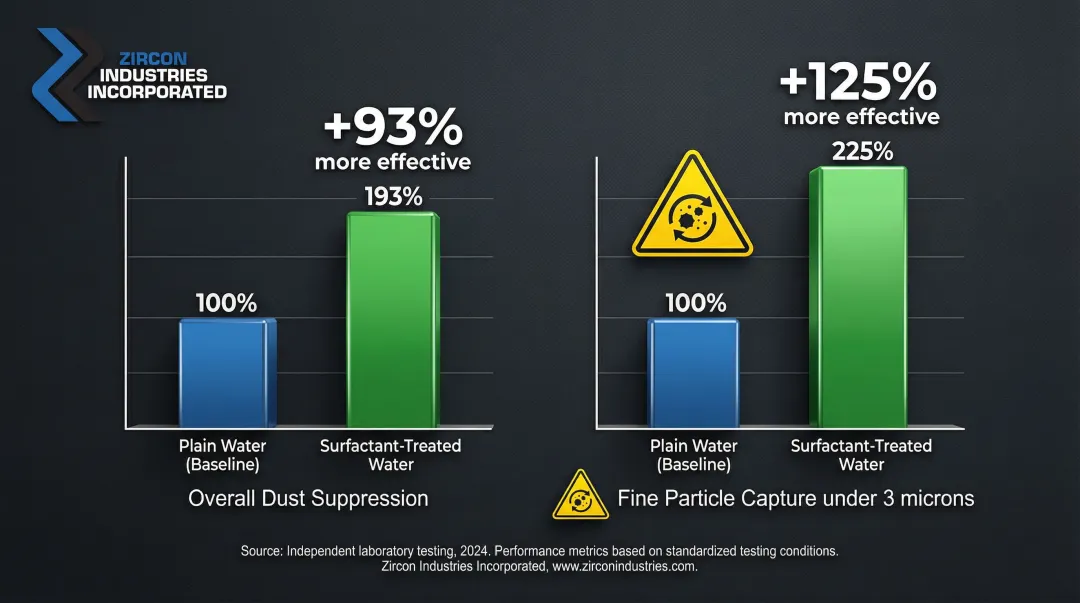
Fine particles below 3 microns are the primary driver of occupational lung disease in coal operations. Those suppression gains translate directly to lower airborne concentrations in breathing zones.
Types of Surfactants Used in Coal Dust Suppression
Nonionic Surfactants: The Workhorses of Coal Dust Control
Nonionic surfactants like Surfynol and Triton X-100 carry no ionic charge on their hydrophilic head group. That means they perform without relying on electrostatic attraction—an important distinction in variable field conditions.
Performance strengths:
- Superior wetting across variable coal types and water conditions
- Not sensitive to water hardness, pH, or dissolved minerals
- Consistently outperform ionic surfactants for weakly-charged or neutralized coal dust aerosols
- Achieve effective hydrophilic-lipophilic balance (HLB) that optimizes coal particle wetting
Research shows nonionic surfactants perform well in capturing weakly-charged coal dust, matching or exceeding ionic spray collection efficiency under real-world conditions where coal dust charge varies throughout the shift.
Adding sodium hydroxide (NaOH) or potassium hydroxide (KOH) to nonionic solutions can improve wetting performance by up to 215%. That combined effect makes alkaline-enhanced nonionic formulations a strong default choice for operations dealing with unpredictable dust charge conditions.
Anionic Surfactants: Targeting Positively-Charged Particles
Anionic surfactants like sodium dodecyl sulfate (SDS) carry a net negative charge on their head group. Their spray droplets attract positively-charged coal dust particles through electrostatic force.
When they work best:
- Coal dust carries strong positive charge (measured by electrostatic monitoring)
- Water chemistry is soft with neutral pH
- Charge characteristics are consistent and predictable
Limitations:
- Underperform for negatively-charged coal dust due to electrostatic repulsion
- Overall mass collection efficiency is lower compared to cationic and nonionic alternatives
- Sensitive to hard water (calcium and magnesium ions interfere with surfactant action)
Cationic Surfactants: For Negatively-Charged Dust Scenarios
Cationic surfactants like dodecylamine hydrochloride (DAH) carry a net positive charge. Their positively-charged spray droplets attract negatively-charged coal dust particles.
When they work best:
- Coal dust consistently carries strong negative charge
- Specific mining operation has verified charge characteristics through testing
- Water chemistry is controlled and compatible
Cautions:
- Sensitive to hard mine water
- May interact negatively with mine surfaces, equipment, or other chemical additives
- Performance advantage only realized when dust charge matches surfactant strategy
Foam Systems: High-Expansion Enhancement
Foam systems apply a surfactant-water mixture as high-expansion foam rather than a liquid spray, dramatically increasing surface contact with airborne dust while cutting water consumption. Studies show foam can provide 20-60% additional dust reduction compared to water sprays alone, using significantly less water.
Documented performance:
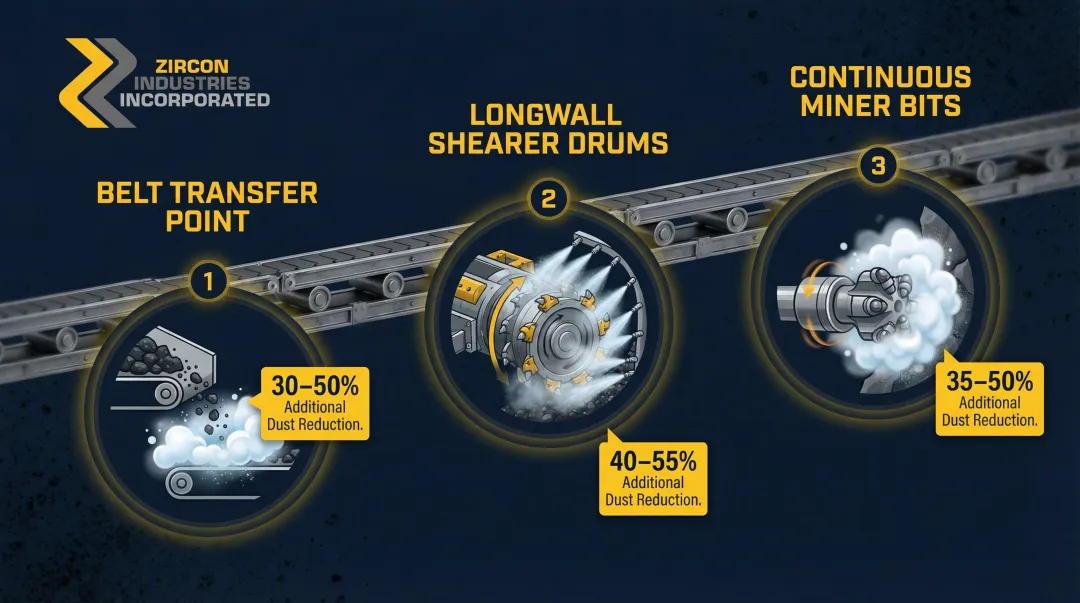
- Seibel (1976) demonstrated 30% additional dust reduction at belt transfer points using high-expansion foam versus water sprays
- Mukherjee & Singh (1984) found foam released from longwall shearer drums cut dust by 50% compared to conventional water sprays, while using only half the water volume
Practical recommendation: In most underground and surface mining environments, coal dust charge varies by location, seam type, and time of shift—making charge-dependent ionic surfactants a gamble without continuous monitoring. Nonionic surfactants sidestep that uncertainty entirely. Ionic options (anionic or cationic) are worth evaluating only when your operation has confirmed stable, strong dust charge through direct electrostatic measurement.

Key Factors That Affect Surfactant Selection and Performance
Coal Type and Sulfur Content
Collection efficiency for different coals is strongly influenced by sulfur content. Higher-sulfur bituminous coals respond differently to surfactant wetting than low-sulfur lignite or anthracite. Coal rank also affects wettability—long flame coal generally exhibits stronger wettability than gas coal.
There is no universal formula. Test candidate surfactants against your specific coal before committing to a program. What works for Pittsburgh seam coal may underperform for Powder River Basin coal.
Water Quality: pH and Mineralogy
Mine water chemistry varies dramatically between operations. Hard water (high calcium and magnesium content) deactivates or reduces the effectiveness of ionic surfactants. Extreme pH levels—whether acidic from sulfur oxidation or alkaline from carbonate minerals—also interfere with ionic surfactant performance.
Nonionic surfactants tolerate variable water conditions better than ionic alternatives, making them the safer choice when water chemistry fluctuates or isn't well-characterized. Adding alkaline base to nonionic solutions can significantly improve wetting performance, though verify compatibility with your specific water chemistry first.
Particle Size Distribution
Surfactant impact is most pronounced for fine particles below 3 microns—the hardest to capture and the most dangerous for lung health. Collection efficiency for coarser particles (>5 microns) is primarily driven by inertial impaction and is less sensitive to surfactant type.
Submicron respirable particles depend heavily on surface tension reduction and charge effects. If your primary concern is meeting the 1.5 mg/m³ respirable dust PEL and your monitoring shows high fine particle counts, prioritize surfactants with proven sub-3 micron capture performance.
Surfactant Concentration: Finding the CMC Sweet Spot
More is not always better. The relationship between surfactant concentration and performance is non-linear. Effectiveness plateaus near the critical micelle concentration (CMC). Beyond this point, additional surfactant molecules form micelles within the solution rather than interacting at the surface—delivering no additional dust capture benefit.
Typical CMC thresholds vary by surfactant type:
- Triton X-100: Optimal efficiency around 0.10% concentration (lower CMC)
- SDS: Peak performance at 0.15–0.20% concentration (higher CMC)
- Excess dosing: No suppression gain—only added cost and product waste
Work with a chemical supplier experienced in mining applications—DirectChem, for example, helps operators identify the right dosage for their specific spray system and coal type—to avoid both under-dosing and costly overuse.
Application Methods and Implementation Best Practices
Water Spray Systems: Conventional and High-Pressure
Conventional low-pressure sprays (used at transfer points, longwall faces, continuous miners) benefit significantly from surfactant addition. These systems typically operate at 40-60 psi and deliver 1-5 gallons per minute (gpm) per nozzle.
High-pressure sprays (80-100 psi) create smaller droplets with higher velocity, improving airborne dust capture per unit of water used. However, pressures exceeding 100 psi entrain large air volumes, causing severe turbulence and "dust rollback" that pushes contaminated air back over operators. The sweet spot for most drum sprays is 80-100 psi.
Nozzle configurations:
- Hollow cone sprays deliver fine droplets in a circular pattern, ideal for general dust suppression zones
- Full cone sprays provide denser coverage for high dust generation points
- Flat fan sprays (1 gpm) work well for anti-rollback systems on continuous miner booms, angled for low reach with no overspray
Wet Scrubbers and Integrated Ventilation Systems
Wet scrubbers pull dust-laden mine air through a water curtain, capturing particles before air recirculates or exhausts. Surfactant-dosed water dramatically improves scrubber efficiency, especially for fine particles.
Two variables drive scrubber performance:
- Pump injection pressure — higher pressure produces finer droplets, increasing the total water surface area available for particle contact
- Surfactant concentration — getting the concentration right is critical. Too little reduces effectiveness; too much wastes product and generates excess foam in recirculation systems
Foam Application at Transfer Points and Shearers
Where recirculation foam is a problem to avoid, deliberately applied foam is a controlled advantage. Foam systems deliver 20-60% greater dust reduction than water sprays while using less water, making them ideal for water-restricted areas or operations where water management is costly.
Application points include:
- Belt transfer points where coal drops from one conveyor to another
- Longwall shearer drums where cutting generates intense dust clouds
- Continuous miner bits where bit friction creates high dust concentrations at the source
Foam expansion ratio (air-to-surfactant-solution ratio) affects performance. High-expansion foams (100:1 or greater) provide excellent coverage but collapse quickly. Medium-expansion foams (20:1 to 50:1) offer better persistence for sustained dust suppression.
Turnkey Chemical Delivery and Application Programs
Mine operators and aggregate producers can partner with chemical suppliers like DirectChem, which manufactures and applies dust control products directly on-site through a turnkey service model. These programs eliminate manual handling errors, ensure consistent dosing through automated systems, and reduce labor costs while maintaining compliance with MSHA and occupational exposure standards.
Turnkey programs typically include:
- Site assessment and surfactant selection testing
- Chemical delivery on scheduled intervals
- Automated dosing system installation and monitoring (where applicable)
- Regulatory compliance documentation and record-keeping support
- Performance monitoring and optimization adjustments
Frequently Asked Questions
What chemicals are used for coal dust suppression?
Surfactants (wetting agents) are the primary chemical additives — nonionic types (Surfynol, Triton X-100), anionic types (sodium dodecyl sulfate), and cationic types (dodecylamine hydrochloride). Added to water spray systems, they reduce surface tension and improve wetting of hydrophobic coal particles. Foam agents and polymer binders supplement these in specialized applications such as transfer points and static pile sealing.
Why are nonionic surfactants generally preferred for coal dust suppression?
Nonionic surfactants perform consistently well across different coal types, water conditions, and dust charge levels because they're not sensitive to water hardness or pH. They achieve an effective hydrophilic-lipophilic balance that optimizes coal wetting. Studies comparing surfactant classes show they outperform ionic types, particularly for weakly-charged or mixed-charge coal dust aerosols—the most common scenario in mining operations.
How does water pH and hardness affect surfactant performance in coal mines?
Ionic surfactants (anionic and cationic) can be deactivated or reduced in effectiveness by hard mine water (high calcium/magnesium content) or extreme pH levels. Nonionic surfactants are more tolerant of variable water chemistry. Operators can also improve nonionic performance by adding small amounts of alkaline base (sodium hydroxide or potassium hydroxide) to the solution, which can boost wetting effectiveness by up to 215%.
What concentration of surfactant should be used in coal dust suppression sprays?
Optimal concentration varies with surfactant type, coal characteristics, and spray system design — exceeding the critical micelle concentration (CMC) wastes money without improving performance. Triton X-100 typically performs best around 0.10%, while SDS requires 0.15–0.20%. On-site testing against supplier technical guidance is the reliable way to confirm the right dosage for your conditions.
Can surfactant sprays suppress both coarse and fine coal dust particles?
Yes, surfactant sprays improve capture of both size ranges, but the effect is most pronounced for fine particles below 3 microns — the most dangerous for lung health and the hardest for plain water to capture. Nonionic surfactants suppress fine dust up to 125% more effectively than plain water alone, while coarse particle capture is less sensitive to surfactant type.
Are there regulatory standards governing coal dust suppression in mining operations?
MSHA (Mine Safety and Health Administration) sets a mandatory permissible exposure limit of 1.5 mg/m³ for respirable coal dust and requires mines to implement effective dust control programs under 30 CFR Parts 70, 71, and 75. These regulations explicitly permit the use of "water or water with a wetting agent added to it" to abate excessive dust. Operators must develop approved ventilation and dust control plans detailing their suppression systems, including surfactant use where applicable.


