
Introduction
Ice bonding occurs when moisture on a pavement surface freezes at or below 32°F, creating a molecular bond between the ice layer and the road. This bond demands significant force or chemical energy to break, turning routine winter maintenance into a costly battle.
For road maintenance crews, townships, and industrial site operators, ice bonding creates dangerous conditions, drives accident rates higher, and forces emergency operations that strain both budgets and crews.
The numbers make the case for getting ahead of it: reactive deicing—waiting until snow accumulates before treating—requires up to four times more chemical than proactive treatment and costs substantially more per lane-mile.
Preventing ice from bonding in the first place (anti-icing) is far more effective and cost-efficient than breaking a bond that has already formed. This guide covers the science behind ice bonding, the conditions that accelerate it, and anti-icing strategies that cut chemical use, labor hours, and road hazards.
TL;DR
- Anti-icing prevents ice from bonding by applying freezing-point depressants before precipitation begins
- Liquid chemical pre-treatment uses 75% less material than reactive deicing
- Apply 12–18 hours before a storm when pavement temperature nears 32°F — never when incoming rain will wash the treatment away
- Automated application systems cut labor costs and deliver consistent chemical coverage with no product waste
Why Ice Bonds to Streets: Understanding the Root Causes
Ice bonding begins when moisture on pavement freezes at or below 32°F, creating a tight chemical bond between the ice layer and the road surface. Breaking that bond demands far more force—and far more chemical—than preventing it from forming.
Four primary conditions contribute to ice bond formation:
Pavement Temperature Drops Below Freezing
Pavement temperature—not air temperature—drives ice bond formation. While air cools and warms quickly, pavement retains heat longer but also stays cold longer once it drops below freezing.
High-risk zones experience faster and more severe cooling:
- Bridge decks and overpasses (exposed on all sides with no ground insulation)
- Shaded road sections where sunlight never reaches
- Areas with poor drainage where moisture accumulates
These zones can remain below 32°F even when air temperatures rise, maintaining the ice bond long after a storm passes.
High Humidity and Dew Point Conditions
Frost and black ice form when pavement surface temperature drops to or below the dew point under high humidity, even without any precipitation. This catches crews off guard because no snow or rain triggers treatment protocols.
Black ice is especially dangerous because it's nearly invisible and forms rapidly under these atmospheric conditions. Drivers encounter ice on what appears to be a dry road, often with no warning signs or reduced speed limits in place.
Delayed or Reactive Treatment Approach
Waiting until snow accumulates before treating allows a compacted snow layer to form a strong bond with pavement. Breaking this bond requires much larger chemical quantities. Field studies show reactive deicing uses up to 400% more material than proactive anti-icing.
Reactionary deicing creates operational delays, overtime labor costs, and elevated safety risk during the window when roads are bonded and untreated. By the time crews respond, accidents may have already occurred and traffic backups formed.
Insufficient Moisture or Wrong Chemical for Conditions
Dry pavement kills activation. Solid chemicals need moisture to dissolve and work. Without it, the material sits on the surface with no freeze-point protection—treatment was applied, but the road is still unprotected.
Using a chemical with an insufficient freeze-point depression range for ambient temperature—such as standard rock salt below -6°F pavement temperature—leaves ice bonding unchecked. The chemical simply doesn't work in those conditions, regardless of application rate.
What Happens When Ice Bonding Is Ignored
Bonded ice drives up accident risk fast, triggering road closures and speed restrictions that stall commerce and delay emergency response. That domino effect forces emergency deicing operations—requiring far more chemical volume and labor hours, typically during overtime when costs peak.
Those disruptions translate directly into budget damage. Reactive deicing reliably costs more than proactive anti-icing, and the gap compounds across four cost drivers:
- Chemical volume (4x higher application rates)
- Labor hours (emergency callouts and overtime)
- Equipment wear (aggressive plowing and scraping)
- Liability exposure (accidents during untreated periods)

Warning Signs Ice Bonding Is Imminent
Several clear conditions signal that ice bonding is about to occur:
- Pavement temperature near 32°F on bridge decks or shaded sections, especially with precipitation in the forecast
- Air temperature at or near the dew point with high humidity—even without precipitation, these conditions produce frost or black ice
- Residual chemical visibly diluted by melt or rain, meaning freeze-point protection has dropped and refreeze is likely
These warning signs give crews a window to act proactively before the bond forms.
How to Prevent Ice from Bonding to Streets: Anti-Icing Solutions
Anti-icing works by placing a chemical freezing-point depressant on the pavement surface before ice can form a bond. The chemical acts as a barrier between the road and falling precipitation, preventing the molecular adhesion that creates dangerous conditions.
Liquid Chemical Pre-Treatment (Anti-Icing Spray Application)
Applying liquid brine (sodium chloride, magnesium chloride, or calcium chloride solution) to the road surface before a storm is the gold standard anti-icing method. Liquids adhere to pavement, don't bounce off like solids, and distribute into tire treads to spread coverage.
Timing is critical: Apply when pavement temperature is dropping toward freezing and precipitation is forecast within 12-18 hours. The Wisconsin DOT recommends this advance window to allow water to evaporate, leaving concentrated chemical residue on the surface.
However, applying too early risks washoff. If the storm begins as rain before transitioning to snow, the chemical washes away completely. FHWA guidance emphasizes that pretreatment should not be made when a storm starts with above-freezing temperatures and rain.
Material efficiency: Liquid anti-icing requires approximately one-quarter the chemical volume of reactive deicing, according to industry research. This translates directly to cost savings—less product purchased, transported, and applied.
Prewetted Solid Chemical Application
Prewetting coats dry solid salt (NaCl) with a liquid chemical (CaCl2 or MgCl2 solution) before spreading. This activates the material faster, reduces bounce-off waste by up to 30%, and improves adhesion to the road surface.
When to use prewetted solids instead of straight liquid:
- Colder temperatures (approximately 23°F and below) where pure liquid application is less effective
- When liquid spray equipment is unavailable or impractical
- When heavier application rates are needed for severe conditions
Industry surveys of 33 state DOTs found that 9 to 12 gallons of liquid per ton of solid salt is the most common prewetting ratio, balancing material retention with operational efficiency.
Strategic Timing and Weather Intelligence
Effective anti-icing depends on treating the road before a strong bond forms—not after accumulation has already begun. Crews should use road weather information systems (RWIS), pavement temperature sensors, and contract weather forecasting to determine the optimal treatment window.
Key timing factors to coordinate:
- Pavement temperature: Treat when surface temps are dropping toward freezing, not after they've stabilized below it
- Storm onset: Confirm precipitation type—rain before snow can wash away pretreatment entirely
- Traffic timing: Complete treatment before peak rush hours, when accidents carry the highest impact
Automated Application Systems
Automated deicer spray systems eliminate human error in application rate control, ensuring a precise, consistent amount of chemical is applied per lane-mile regardless of truck speed. This reduces both waste from over-application and the under-treatment that leaves sections vulnerable.
The chemical formulation used matters as much as the delivery system. Freeze control products need to perform at low temperatures — some formulations rated to -50°F or below — to remain effective through the full winter season. DirectChem's freeze control chemical line is formulated for demanding road maintenance applications, including pretreatment and anti-icing programs run by townships and road crews managing large route networks.

Choosing the Right Anti-Icing Chemical for Your Conditions
No single chemical is ideal for every condition. The right choice depends on pavement temperature range, precipitation type, budget, environmental sensitivity, and available equipment.
Sodium Chloride Brine (Salt Brine)
NaCl brine is the most widely used anti-icing liquid: effective to approximately -6°F, cost-effective, easy to produce on-site, and well-documented for highway use. FHWA guidance indicates that application rates are given only for pavement temperatures at 23°F and above because of ineffectiveness below this threshold.
Best for moderate winter climates where pavement temperatures rarely drop below single digits.
Calcium Chloride (CaCl2)
CaCl2 offers distinct advantages:
- Hygroscopic (absorbs moisture from air, activating even on dry pavement)
- Exothermic (releases heat when dissolving, accelerating ice melting)
- Effective to -20°F in liquid form
- Faster-acting than sodium chloride
This makes calcium chloride valuable in colder conditions or as a prewetting agent for solid salt applications. Higher material cost and increased corrosivity compared to NaCl are the main drawbacks.
Magnesium Chloride (MgCl2)
MgCl2 is widely used by DOTs for pre-wetting salt and as a standalone anti-icer. Key characteristics:
- Effective to -10°F in liquid form
- Less corrosive than calcium chloride
- **Applied as a 30% solution** in most field operations
- More cost-effective than CaCl2 in most markets
Minnesota DOT guidelines recommend applying MgCl2 when pavement temperatures range from -10°F to 30°F, making it a versatile mid-range option.
Acetate-Based and Specialty Chemicals
Potassium acetate (KAc) and calcium magnesium acetate (CMA) are lower-corrosion, environmentally preferable options used for sensitive areas:
- Bridge decks (where corrosion impacts structural steel)
- Airport pavements (where chlorides damage aircraft components)
- Near water bodies (where chloride runoff harms aquatic ecosystems)
The trade-off is significantly higher cost—often 3-5 times more expensive than chloride-based products.
Chemical Comparison at a Glance
| Chemical | Effective To | Relative Cost | Best Use Case |
|---|---|---|---|
| Sodium Chloride Brine | -6°F | Low | Moderate climates, highway routes |
| Calcium Chloride | -20°F | Medium-High | Extreme cold, fast activation needed |
| Magnesium Chloride | -10°F | Medium | Versatile mid-range, DOT pre-wetting |
| Potassium Acetate / CMA | Varies | High | Bridges, airports, sensitive environments |
Tips for Building a Long-Term Anti-Icing Program
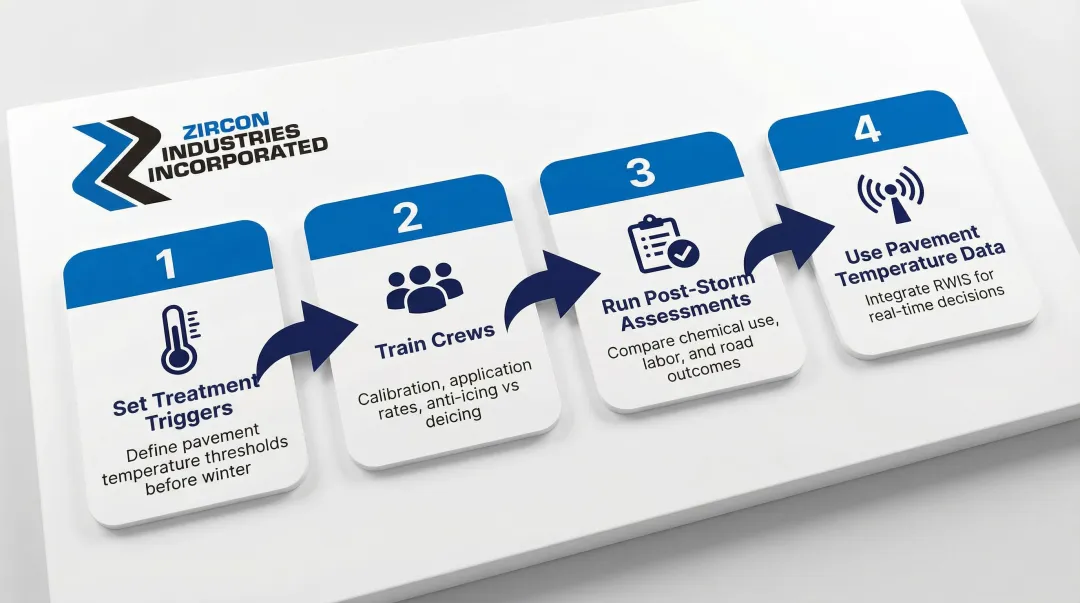
A systematic, season-long anti-icing program runs on consistent protocols and data — not guesswork. Before the first storm hits, lock in your procedures across four areas:
- Set treatment triggers early. Define pavement temperature thresholds and storm response criteria before winter begins. Document them in written protocols every operator can access.
- Train crews on the full picture. Cover correct application rates, equipment calibration, and the distinction between anti-icing and deicing. Inconsistent training leaves some routes over-treated and others under-protected.
- Run post-storm assessments. After each weather event, compare chemical usage, labor hours, and road condition outcomes. Apply those findings to refine timing and rates for the next storm — precipitation type and temperature range both affect what works.
- Use pavement temperature data. Integrate RWIS sensors or pavement monitoring tools into your decision workflow. Air temperature alone doesn't tell the full story; pavement temperature is what actually determines whether ice will bond.
Frequently Asked Questions
What is the difference between anti-icing and deicing?
Anti-icing is applied before ice bonds to pavement to prevent bond formation, while deicing is applied after a bond has already formed to break it. Deicing requires more chemical, more labor, and more time to achieve road clearance.
When should anti-icing chemicals be applied to roads?
Apply 12-18 hours before precipitation begins, while pavement temperature is dropping toward freezing but before snow or ice has accumulated and bonded. Avoid application when rain is forecast to arrive before freezing precipitation — it will wash off the chemical before it can work.
What liquid is sprayed on roads before a snowstorm?
Salt brine (sodium chloride solution) is the most commonly used anti-icing spray, effective to approximately -6°F pavement temperature. Calcium chloride and magnesium chloride solutions are also widely used, especially in colder conditions below 20°F.
Does anti-icing work in extreme cold temperatures?
Standard sodium chloride brine loses effectiveness below approximately -6°F pavement temperature. For colder applications, calcium chloride (effective to -20°F) or specialty acetate-based chemicals are used. Chemical selection is critical for extreme cold performance.
How much can anti-icing reduce road maintenance costs compared to deicing?
Anti-icing uses roughly 25% of the chemical volume required for reactive deicing. Additional savings come from reduced labor hours, eliminated overtime, and fewer accident-related liabilities during untreated periods.
Can anti-icing be used on private roads, mine haul roads, or industrial sites?
Anti-icing is applicable beyond public highways—including mine haul roads, quarry access roads, and industrial sites where bonded ice creates equipment downtime and safety hazards. The same chemical principles and application methods apply, with automated spray systems particularly valuable for large private road networks.


